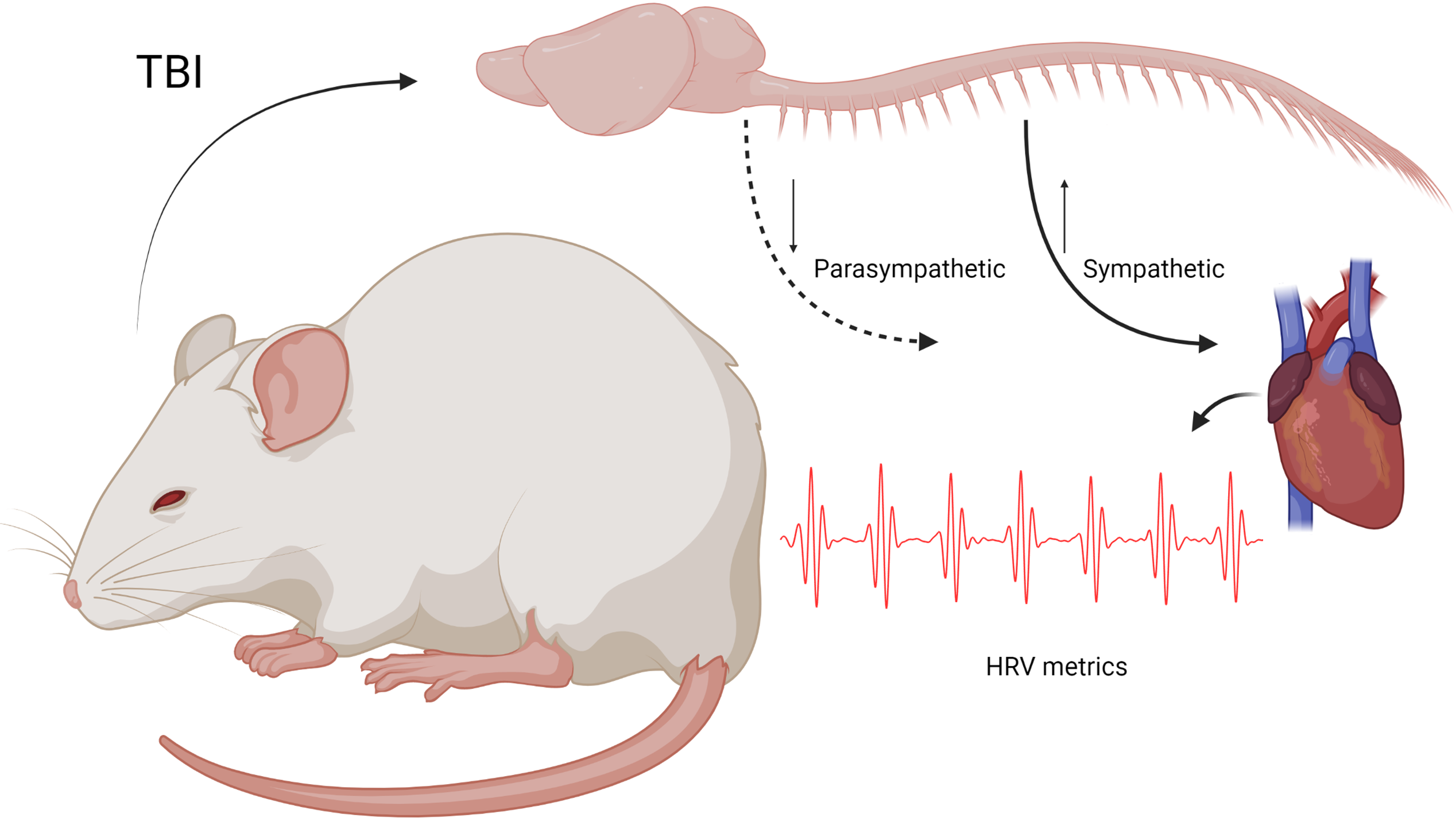
Influence of Traumatic Brain Injury by Fluid Percussion on Heart Rate Variability in the Acute Phase of Damage in Rats
Keywords:
Fluid percussion injury, Heart rate variability, Autonomic nervous system, Electrocardiogram, Isoflurane anesthesiaAbstract
Traumatic brain injury (TBI) is a condition that changes the autonomic system, modulating the heart rate variability (HRV) at all levels of brain lesions. Although fluid percussion injury (FPI) model can reproduce all degrees of severity of clinical TBI, there is still a lack of comprehensive analysis of linear and non-linear HRV metrics following FPI. The present study sought to assess the influence of the FPI model on time-domain (HR, mean NN, SD1, SD2, SDNN, RMSSD, and SD1/SD2) and frequency-domain (LF, HF, and LF/HF). A non-invasive electrocardiogram recording was used in anesthetized and awake male Wistar rats, both before and for three days after moderate FPI. Although a decrease in the SD2 occurred in the anesthetized state, an increase in HFnu led to a reduction in HR during baseline evaluations. Post-TBI analyses revealed that neither the sham nor the TBI groups exhibited HR alterations under the influence of isoflurane; however, both groups showed a decrease in parasympathetic activity (RMSSD, SD1, and HFnu). Under isoflurane anesthesia, only the TBI group exhibited changes in LFnu, HFnu, and LF/HF metrics for three days. In contrast, awake animals experienced an increase in HR for three days post-injury, with a critical period at 24 hours when SD2, LFnu, HFnu, and LF/HF were altered. With few exceptions, the sham group did not exhibit significant differences in the awake state. Therefore, the effects of isoflurane predominate over TBI effects in both time- and frequency-domain metrics, while FPI in awake animals indicates a critical period of altered specific metrics at 24 hours post-injury.
Downloads
References
H.-Y. Lu, A. P.-H. Huang, and L.-T. Kuo, “Prognostic value of variables derived from heart rate variability in patients with traumatic brain injury after decompressive surgery,” PLoS One, vol. 16, no. 2, p. e0245792, 2021, doi: 10.1371/journal.pone.0245792.
L. F. F. Royes and F. Gomez-Pinilla, “Making sense of gut feelings in the traumatic brain injury pathogenesis,” Neurosci Biobehav Rev, vol. 102, pp. 345-361, 2019, doi:10.1016/j.neubiorev.2019.05.012.
R. L. Conder and A. A. Conder, “Heart rate variability interventions for concussion and rehabilitation,” Frontiers in Psychology, vol. 5, no. 890, 2014, doi: 10.3389/fpsyg.2014.00890.
S. J. McDonald et al., “Beyond the brain: peripheral interactions after traumatic brain injury,” J Neurotrauma, vol. 37, no. 5, pp. 770–781, 2020, doi: 10.1089/neu.2019.6885.
J. P. Abaji, D. Curnier, R. D. Moore, and D. Ellemberg, “Persisting effects of concussion on heart rate variability during physical exertion,” J Neurotrauma, vol. 33, no. 9, pp. 811–817, 2016, doi: 10.1089/neu.2015.3989.
M. Hasen, A. Almojuela, and F. A. Zeiler, “Autonomic dysfunction and associations with functional and neurophysiologic outcome in moderate / severe traumatic brain injury : a scoping review,” J Neurotrauma, vol. 36, no. 10, pp. 1–56, 2019, doi: 10.1089/neu.2018.6073.
M. Sykora et al., “Autonomic impairment in severe traumatic brain injury: A multimodal neuromonitoring study,” Crit Care Med, vol. 44, no. 6, pp. 1173–1181, 2016, doi: 10.1097/CCM.0000000000001624.
D. I. Graham, T. K. McIntosh, W. L. Maxwell, and J. A. R. Nicoll, "Recent advances in neurotrauma," J. Neuropathol. Exp. Neurol., vol. 59, no. 8, pp. 641–651, 2000, doi: 10.1093/jnen/59.8.641.
M. Albrecht, J. Henke, S. Tacke, M. Markert, and B. Guth, “Effects of isoflurane , ketamine-xylazine and a combination of medetomidine , midazolam and fentanyl on physiological variables continuously measured by telemetry in Wistar rats,” BMC Vet Res, vol. 10, p. 198, 2014, doi: 10.1186/s12917-014-0198-3.
S. Fenske et al., “Comprehensive multilevel in vivo and in vitro analysis of heart rate fluctuations in mice by ECG telemetry and electrophysiology,” Nat Protoc, vol. 11, no. 1, pp. 61–86, 2016, doi: 10.1038/nprot.2015.139.
I. J. Hildebrandt, H. Su, and W. A. Weber, “Anesthesia and other considerations for in vivo imaging of small animals,” ILAR J, vol. 49, no. 1, pp. 17–26, 2008, doi: 10.1093/ilar.49.1.17.
R. S. Nascimento, F. S. Fiorin, A. R. S. Santos, L. F. F. Royes, and J. L. B. Marques, “A new approach for ECG recording in rats: an autonomic nervous system analysis,” in Proceedings of the 5th Brazilian Technology Symposium, Smart Innovation, Systems and Technologies 202, Springer Nature Switzerland, 2020, doi: 10.1007/978-3-030-57566-3_9.
A. Bilan, A. Witczak, R. Palusiński, W. Myśliński, and J. Hanzlik, “Circadian rhythm of spectral indices of heart rate variability in healthy subjects,” J. Electrocardiol., vol. 38, no. 3, pp. 239–243, 2005, doi: 10.1016/j.jelectrocard.2005.01.012.
R. S. Nascimento, J. L. Brum Marques, A. R. Soares Santos, L. F. Freire Royes, and F. da Silva Fiorin, “Development and application of a novel pressure system for evaluating trauma severities using a physiological approach after traumatic brain injury in rats,” World Neurosurg, vol. 177, pp. e354–e360, Sep. 2023, doi: 10.1016/j.wneu.2023.06.049.
R. S. Nascimento, F. Silva Fiorin, and J. L. B. Marques, “Design and implementation of an ECG recording system for in Vivo experimentation in rats,” in Smart Innovation, Systems and Technologies, vol. 233, 2021, pp. 334–341. doi: 10.1007/978-3-030-75680-2_37.
Task Force of the European Society of Cardiology and The North American Society of Pacing and Electrophysiology, “Heart rate variability: standards of measurement, physiological interpretation, and clinical use,” Eur Heart J, vol. 17, no. 3, pp. 354–381, 1996, doi: 10.1161/01.CIR.93.5.1043.
F. Shaffer, J. P. Ginsberg, “An overview of heart rate variability metrics and norms,” Front. Public Health, vol. 5, pp. 258, 2017, doi: 10.3389/fpubh.2017.00258.
M. P. Tulppo, T. H. Mäkikallio, T. E. S. Takala, T. Seppanen, and H. V Huikuri, “Quantitative beat-to-beat analysis of heart rate dynamics during exercise,” Am J Physiol, vol. 271, no. 1 pt 2, pp. H244–H252, 1996, doi: 10.1152/ajpheart.1996.271.1.H244.
W. H. 3rd Rowan, M. J. Campen, L. B. Wichers, and W. P. Watkinson, “Heart rate variability in rodents: uses and caveats in toxicological studies,” Cardiovasc Toxicol, vol. 7, no. 1, pp. 28–51, 2007, doi: 10.1007/s12012-007-0004-6.
P. Melillo et al., “Automatic prediction of cardiovascular and cerebrovascular events using heart rate variability analysis,” PLoS One, vol. 10, no. 3, p. e0118504, 2015, doi: 10.1371/journal.pone.0118504.
C. Su, T. B. Kuo, J. Kuo, H. Lai, and H. I. Chen, “Sympathetic and parasympathetic activities evaluated by heart-rate variability in head injury of various severities *,” Clin Neurophysiol, vol. 116, no. 6, pp. 1273–1279, 2005, doi: 10.1016/j.clinph.2005.01.010.
L. Carnevali and A. Sgoifo, “Vagal modulation of resting heart rate in rats : the role of stress, psychosocial factors, and physical exercise,” Front Physiol, vol. 5, p. 118, 2014, doi: 10.3389/fphys.2014.00118.
L. J. Mercier et al., “Altered autonomic cardiovascular function in adults with persisting post‐concussive symptoms and exercise intolerance, ” Physiol. Rep., vol. 13, no. 11, p. e70378, 2025, doi: 10.14814/phy2.70378.
A. B. Stein et al., “Effects of anesthesia on echocardiographic assessment of left ventricular structure and function in rats,” Basic Res Cardiol, vol. 102, no. 1, pp. 28–41, 2007, doi: 10.1007/s00395-006-0627-y.
R. E. Pachon, B. A. Scharf, D. E. Vatner, and S. F. Vatner, “Best anesthetics for assessing left ventricular systolic function by echocardiography in mice,” Am J Physiol Heart Circ Physiol, vol. 308, no. 12, pp. H1525–H1529, 2015, doi: 10.1152/ajpheart.00890.2014.
C. Sévoz-Couche et al., “Involvement of the dorsomedial hypothalamus and the nucleus tractus solitarii in chronic cardiovascular changes associated with anxiety in rats,” J Physiol, vol. 591, no. 7, pp. 1871–1887, 2013, doi: 10.1113/jphysiol.2012.247791.
S. K. Wood, K. V Mcfadden, D. Grigoriadis, S. Bhatnagar, and R. J. Valentino, “Depressive and cardiovascular disease comorbidity in a rat model of social stress: a putative role for corticotropin-releasing factor,” Psychopharmacology (Berl), vol. 222, no. 2, pp. 325–336, 2012, doi: 10.1007/s00213-012-2648-6.
C. Lerma, O. Infante, H. Pérez-Grovas, and M. V José, “Poincaré plot indexes of heart rate variability capture dynamic adaptations after haemodialysis in chronic renal failure patients,” Clin Physiol Funct Imaging, vol. 23, no. 2, pp. 72–80, 2003, doi: 10.1046/j.1475-097x.2003.00466.x.
C. C. Grant, C. Murray, D. C. Janse van Rensburg, and L. Fletcher, “A comparison between heart rate and heart rate variability as indicators of cardiac health and fitness,” Front Physiol, vol. 4, p. 337, 2013, doi: 10.3389/fphys.2013.00337.
C. H. Hsu et al., “Poincaré plot indexes of heart rate variability detect dynamic autonomic modulation during general anesthesia induction,” Acta Anaesthesiol Taiwan, vol. 50, no. 1, pp. 12–18, Mar. 2012, doi: 10.1016/j.aat.2012.03.002.
V. Krishnamoorthy et al., “Association of early hemodynamic profile and the development of systolic dysfunction following traumatic brain injury,” Neurocrit Care, vol. 26, no. 3, pp. 379–387, 2017, doi: 10.1007/s12028-016-0335-x.
A. Deepika et al., “Neuroimmunology of traumatic brain injury: a longitudinal exploratory study,” J. Neuroimmunol, vol. 317, pp. 1–12, 2018, doi: 10.1089/neu.2017.5151.