Attention Blocks Improve White Matter Hyperintensity Semantic Segmentation using U-Nets
Keywords:
attention blocks, artificial intelligence, alzheimer's disease, semantic segmentation, white matter hyperintensitiesAbstract
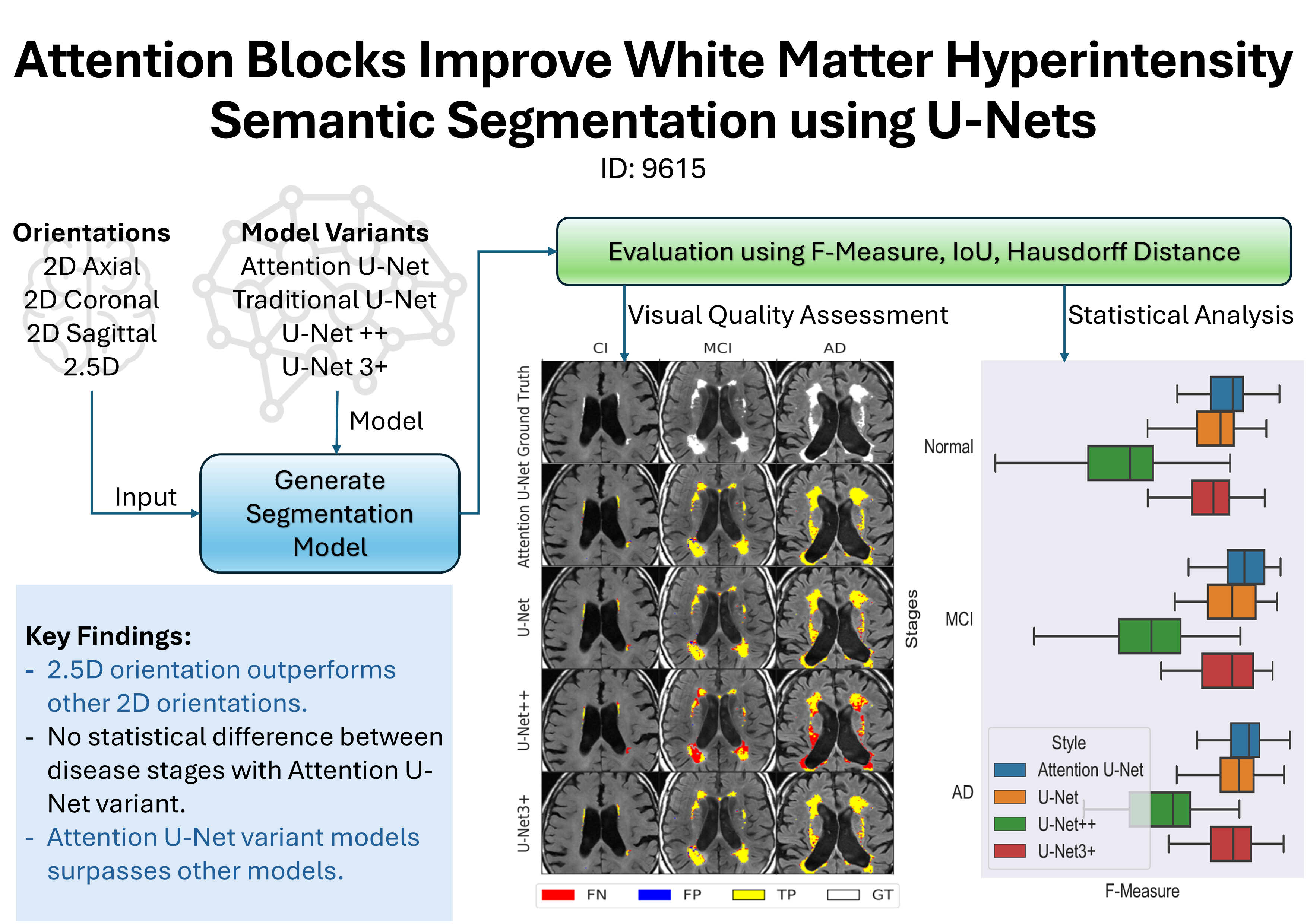
White matter hyperintensities (WMHs) are a common finding on magnetic resonance (MR) images in older individuals, appearing as high-signal intensity regions on fluid-attenuated inversion recovery (FLAIR) imaging. People with high WMH volume are at increased risk for dementia and stroke, controlling for vascular risk factors, but WMH burden is not reliably assessed in clinical practice. Manual segmentation of WMHs is accepted as the gold standard (or ground truth), however, it is a laborious and time-consuming method. Newer machine learning (ML)-based approaches are being proposed as alternatives to manual segmentation. Among these approaches, U-Net convolutional neural networks have demonstrated good WMH segmentation performance. However, even state-of-the-art ML models sometimes fail to correctly identify WMHs and their boundaries with sufficient accuracy. Attention blocks have emerged as a potential solution for improving the performance of U-Net models by enhancing the ability of the model to focus on relevant features in the data. We investigated the effectiveness of attention blocks in U-Net models for WMH segmentation compared to three other models (U-Net++, U-Net3+, and a standard U-Net). Attention blocks significantly improved the F-measure score for WMH segmentation (0.811 vs 0.789 for next best model, p=0.04) in a diverse brain imaging dataset. This study demonstrates that attention blocks enhance U-Net models used for WMH identification and classification.
Downloads
References
J. M. Wardlaw, M. C. V. Hern´andez, and S. Mu˜noz-Maniega, “What are white matter hyperintensities made of?” Journal of the American Heart Association, vol. 4, no. 6, p. e001140, 2015. [Online]. Available:
https://doi.org/10.1161/JAHA.114.001140
C. E. Bauer, V. Zachariou, E. Seago, and B. T. Gold, “White
matter hyperintensity volume and location: Associations with wm
microstructure, brain iron, and cerebral perfusion,” Frontiers in
Aging Neuroscience, vol. 13, 2021. [Online]. Available: https:
//doi.org/10.3389/fnagi.2021.617947
J. M. Wardlaw, E. E. Smith, G. J. Biessels, C. Cordonnier, F. Fazekas,
R. Frayne, R. I. Lindley, J. T. O’Brien, F. Barkhof, O. R. Benavente,
S. E. Black, C. Brayne, M. Breteler, H. Chabriat, C. DeCarli,
F.-E. de Leeuw, F. Doubal, M. Duering, N. C. Fox, S. Greenberg,
V. Hachinski, I. Kilimann, V. Mok, R. van Oostenbrugge, L. Pantoni,
O. Speck, B. C. M. Stephan, S. Teipel, A. Viswanathan, D. Werring,
C. Chen, C. Smith, M. van Buchem, B. Norrving, P. B. Gorelick,
and M. Dichgans, “Neuroimaging standards for research into small
vessel disease and its contribution to ageing and neurodegeneration,”
The Lancet Neurology, vol. 12, no. 8, pp. 822–838, 2013. [Online].
Available: https://doi.org/10.1016/S1474-4422(13)70124-8
C. Haffner, R. Malik, and M. Dichgans, “Genetic factors in cerebral
small vessel disease and their impact on stroke and dementia,” Journal
of Cerebral Blood Flow & Metabolism, vol. 36, no. 1, pp. 158–171,
[Online]. Available: https://doi.org/10.1038/jcbfm.2015.71
K. T. N. Duarte, A. S. Sidhu, M. C. Barros, D. G. Gobbi, C. R.
McCreary, F. Saad, R. Camicioli, E. E. Smith, M. P. Bento, and
R. Frayne, “Multi-stage semi-supervised learning enhances white matter
hyperintensity segmentation,” Frontiers in Computational Neuroscience,
vol. 18, 2024. [Online]. Available: https://doi.org/10.3389/fncom.2024.
K. Duarte, P. V. de Paiva, P. Martins, and M. Carvalho, “Predicting
the early stages of the Alzheimer’s disease via combined brain
multi-projections and small datasets,” in Proceedings of the 14th
International Joint Conference on Computer Vision, Imaging and
Computer Graphics Theory and Applications. SCITEPRESS -
Science and Technology Publications, 2019. [Online]. Available:
http://doi.org/10.5220/0007404705530560
G. Tosto, M. E. Zimmerman, J. L. Hamilton, O. T. Carmichael,
and A. M. Brickman, “The effect of white matter hyperintensities
on neurodegeneration in mild cognitive impairment,” Alzheimer’s and
Dementia, vol. 11, no. 12, pp. 1510–1519, Jun. 2015. [Online].
Available: https://doi.org/10.1016/j.jalz.2015.05.014
S. Lee, F. Viqar, M. E. Zimmerman, A. Narkhede, G. Tosto, T. L.
Benzinger, D. S. Marcus, A. M. Fagan, A. Goate, N. C. Fox, N. J.
Cairns, D. M. Holtzman, V. Buckles, B. Ghetti, and E. M. et al,
“White matter hyperintensities are a core feature of alzheimer’s disease:
Evidence from the dominantly inherited Alzheimer network,” Annals of
Neurology, vol. 79, no. 6, pp. 929–939, Apr. 2016. [Online]. Available:
https://doi.org/10.1002/ana.24647
E. E. Smith, M. O’Donnell, G. Dagenais, S. A. Lear, A. Wielgosz,
M. Sharma, P. Poirier, G. Stotts, S. E. Black, S. Strother, M. D.
Noseworthy, O. Benavente, J. Modi, M. Goyal, S. Batool, K. Sanchez,
V. Hill, C. R. McCreary, R. Frayne, S. Islam, J. DeJesus, S. Rangarajan,
K. Teo, S. Yusuf, and on behalf of the PURE Investigators, “Early
cerebral small vessel disease and brain volume, cognition, and gait,”
Annals of Neurology, vol. 77, no. 2, pp. 251–261, 2015. [Online].
Available: https://doi.org/10.1002/ana.24320
E. Shelhamer, J. Long, and T. Darrell, “Fully convolutional networks
for semantic segmentation,” IEEE Transactions on Pattern Analysis
and Machine Intelligence, vol. 39, no. 4, pp. 640–651, 2017. [Online].
Available: https://doi.org/10.1109/TPAMI.2016.2572683
N. Siddique, S. Paheding, C. P. Elkin, and V. Devabhaktuni, “U-Net and
its variants for medical image segmentation: A review of theory and
applications,” IEEE Access, vol. 9, pp. 82 031–82 057, 2021. [Online].
Available: https://doi.org/10.1109/ACCESS.2021.3086020
O. Ronneberger, P. Fischer, and T. Brox, “U-Net: Convolutional
networks for biomedical image segmentation,” in Medical Image
Computing and Computer-Assisted Intervention – MICCAI 2015,
N. Navab, J. Hornegger, W. M. Wells, and A. F. Frangi, Eds.
Cham: Springer International Publishing, 2015, pp. 234–241. [Online].
Available: https://doi.org/10.48550/arXiv.1505.04597
B. Chen, Y. Liu, Z. Zhang, G. Lu, and A. W. K. Kong,
“Transattunet: Multi-level attention-guided U-Net with transformer
for medical image segmentation,” 2022. [Online]. Available: https:
//doi.org/10.1109/TETCI.2023.3309626
A. Vaswani, N. Shazeer, N. Parmar, J. Uszkoreit, L. Jones, A. N.
Gomez, L. Kaiser, and I. Polosukhin, “Attention is all you need,” 2017.
[Online]. Available: https://doi.org/10.48550/arXiv.1706.03762
R. Heinen, M. D. Steenwijk, F. Barkhof, J. M. Biesbroek, W. M.
van der Flier, H. J. Kuijf, N. D. Prins, H. Vrenken, G. J. Biessels,
J. de Bresser, and TRACE-VCI study group, “Performance of five
automated white matter hyperintensity segmentation methods in a
multicenter dataset,” Scientific Reports, vol. 9, no. 1, 2019. [Online].
Available: https://doi.org/10.1038/s41598-019-52966-0
W. Zhu, H. Huang, Y. Zhou, F. Shi, H. Shen, R. Chen, R. Hua,
W. Wang, S. Xu, and X. Luo, “Automatic segmentation of white
matter hyperintensities in routine clinical brain MRI by 2D VB-Net:
A large-scale study,” Frontiers in Aging Neuroscience, vol. 14, 2022.
[Online]. Available: https://doi.org/10.3389/fnagi.2022.915009
S. Liu, X. Wu, S. He, X. Song, F. Shang, and X. Zhao, “Identification
of white matter lesions in patients with acute ischemic lesions using
U-Net,” Frontiers in Neurology, vol. 11, 2020. [Online]. Available:
https://doi.org/10.3389/fneur.2020.01008
V. Sundaresan, G. Zamboni, P. M. Rothwell, M. Jenkinson,
and L. Griffanti, “Triplanar ensemble U-Net model for white
matter hyperintensities segmentation on MR images,” Medical Image
Analysis, vol. 73, p. 102184, 2021. [Online]. Available: https:
//doi.org/10.1016/j.media.2021.102184
G. Park, J. Hong, B. A. Duffy, J.-M. Lee, and H. Kim, “White matter
hyperintensities segmentation using the ensemble U-Net with multiscale
highlighting foregrounds,” NeuroImage, vol. 237, p. 118140, 2021.
[Online]. Available: https://doi.org/10.1016/j.neuroimage.2021.118140
S. Lee, Z. Rieu, R. E. Kim, M. Lee, K. Yen, J. Yong, and D. Kim,
“Automatic segmentation of white matter hyperintensities in T2-FLAIR
with AQUA: A comparative validation study against conventional
methods,” Brain Research Bulletin, vol. 205, p. 110825, 2023.
[Online]. Available: https://www.sciencedirect.com/science/article/pii/
S0361923023002502
H. J. Kuijf, A. Casamitjana, D. L. Collins, M. Dadar, A. Georgiou, and
et al., “Standardized assessment of automatic segmentation of white
matter hyperintensities and results of the WMH segmentation challenge,”
IEEE Transactions on Medical Imaging, vol. 38, pp. 2556–2568, 2019.
[Online]. Available: https://doi.org/10.1109/tmi.2019.2905770
K. T. Duarte, D. G. Gobbi, A. S. Sidhu, C. R. McCreary, F. Saad,
R. Camicioli, E. E. Smith, and R. Frayne, “Segmenting white matter
hyperintensities in brain magnetic resonance images using convolution
neural networks,” Pattern Recognition Letters, vol. 175, pp. 90–94,
[Online]. Available: https://doi.org/10.1016/j.patrec.2023.07.014
K. T. Duarte, D. G. Gobbi, A. S. Sidhu, C. R. McCreary,
F. Saad, N. Das, E. E. Smith, and R. Frayne, “Segmenting white
matter hyperintensity in Alzheimer’s disease using U-Net cnns,”
in 2022 35th SIBGRAPI Conference on Graphics, Patterns and
Images (SIBGRAPI), vol. 1, 2022, pp. 109–114. [Online]. Available:
https://doi.org/10.1109/SIBGRAPI55357.2022.9991752
C. R. McCreary, M. Salluzzi, L. B. Andersen, D. Gobbi, L. Lauzon,
F. Saad, E. E. Smith, and R. Frayne, “Calgary Normative Study:
Design of a prospective longitudinal study to characterise potential
quantitative MR biomarkers of neurodegeneration over the adult
lifespan,” BMJ Open, vol. 10, no. 8, 2020. [Online]. Available:
https://doi.org/10.1136/bmjopen-2020-038120
S. Peca, C. R. McCreary, E. Donaldson, G. Kumarpillai, N. Shobha, and
et al, “Neurovascular decoupling is associated with severity of cerebral
amyloid angiopathy,” Neurology, vol. 81, no. 19, pp. 1659–1665, 2013.
[Online]. Available: https://doi.org/10.1212/01.wnl.0000435291.49598.
M. S. Albert, S. T. DeKosky, D. Dickson, B. Dubois, H. H. Feldman,
N. C. Fox, A. Gamst, D. M. Holtzman, W. J. Jagust, R. C. Petersen,
P. J. Snyder, M. C. Carrillo, B. Thies, and C. H. Phelps, “The
diagnosis of mild cognitive impairment due to Alzheimer’s disease:
Recommendations from the National Institute on Aging-Alzheimer’s
Association workgroups on diagnostic guidelines for Alzheimer’s
disease,” Alzheimer’s & Dementia, vol. 7, no. 3, pp. 270–279, 2011.
[Online]. Available: https://doi.org/10.1016/j.jalz.2011.03.008
A. Subotic, C. McCreary, F. Saad, A. Nguyen, A. Alvarez-Veronesi,
and et al, “Cortical thickness and its association with clinical cognitive
and neuroimaging markers in cerebral amyloid angiopathy,” Journal of
Alzheimer’s Disease, vol. 81, pp. 1–9, 05 2021. [Online]. Available:
https://doi.org/10.3233/JAD-210138
D. Gobbi, Q. Lu, R. Frayne, and M. Salluzzi, “Cerebra-WML: A rapid
workflow for quantification of white matter hyperintensities,” Canadian
Stroke Congress, vol. 40, pp. E128–E129, 2012. [Online]. Available:
https://www.researchgate.net/publication/278294844 Cerebra-WML a
rapid workflow for quantification of white matter hyperintensities
J. C. Kosior, S. Idris, D. Dowlatshahi, M. Alzawahmah, M. Eesa, and
et al, “Quantomo: Validation of a computer-assisted methodology for
the volumetric analysis of intracerebral haemorrhage,” International
Journal of Stroke, vol. 6, no. 4, pp. 302–305, 2011. [Online]. Available:
https://doi.org/10.1111/j.1747-4949.2010.00579.x
N. J. Tustison, B. B. Avants, P. A. Cook, Y. Zheng, A. Egan, P. A.
Yushkevich, and J. C. Gee, “N4ITK: improved N3 bias correction,”
IEEE Trans. Medical Imaging, vol. 29, no. 6, pp. 1310–1320, 2010.
[Online]. Available: https://doi.org/10.1109/TMI.2010.2046908
K. T. N. Duarte, M. C. de Barros, D. G. Gobbi, A. S. Sidhu, M. A. G.
de Carvalho, and R. Frayne, “Changes in 3D radiomic texture descriptors
in Alzheimer’s disease stages,” in 18th International Symposium on
Medical Information Processing and Analysis, J. Brieva, P. Guevara,
N. Lepore, M. G. Linguraru, L. Rittner, and E. R. C. M.D., Eds., vol.
, International Society for Optics and Photonics. SPIE, 2023, p.
S. [Online]. Available: https://doi.org/10.1117/12.2670246
K. T. N. Duarte, D. G. Gobbi, R. Frayne, and M. A. G.
de Carvalho, “Detecting Alzheimer’s disease based on structural region
analysis using a 3D shape descriptor,” in 2020 33rd SIBGRAPI
Conference on Graphics, Patterns and Images (SIBGRAPI), 2020, pp.
–187. [Online]. Available: https://doi.org/10.1109/SIBGRAPI51738.
00032
R. Guerrero, C. Qin, O. Oktay, C. Bowles, L. Chen, and et al.,
“White matter hyperintensity and stroke lesion segmentation and
differentiation using convolutional neural networks,” NeuroImage:
Clinical, vol. 17, pp. 918–934, 2018. [Online]. Available: https:
//doi.org/10.1016/j.nicl.2017.12.022
O. Oktay, J. Schlemper, L. L. Folgoc, M. Lee, M. Heinrich, K. Misawa,
K. Mori, S. McDonagh, N. Y. Hammerla, B. Kainz, B. Glocker, and
D. Rueckert, “Attention U-Net: Learning where to look for the pancreas,”
[Online]. Available: https://doi.org/10.48550/arXiv.1804.03999
K. Simonyan and A. Zisserman, “Very deep convolutional networks for
large-scale image recognition,” arXiv 1409.1556, 09 2014. [Online].
Available: https://doi.org/10.48550/arXiv.1409.1556
Z. Zhou, M. M. Rahman Siddiquee, N. Tajbakhsh, and J. Liang,
“Unet++: A nested U-Net architecture for medical image segmentation,”
in Deep Learning in Medical Image Analysis and Multimodal Learning
for Clinical Decision Support, D. Stoyanov, Z. Taylor, G. Carneiro,
T. Syeda-Mahmood, A. Martel, L. Maier-Hein, J. M. R. Tavares,
A. Bradley, J. P. Papa, V. Belagiannis, J. C. Nascimento, Z. Lu,
S. Conjeti, M. Moradi, H. Greenspan, and A. Madabhushi, Eds. Cham:
Springer International Publishing, 2018, pp. 3–11. [Online]. Available:
https://doi.org/10.48550/arXiv.1807.10165
H. Huang, L. Lin, R. Tong, H. Hu, Q. Zhang, Y. Iwamoto,
X. Han, Y.-W. Chen, and J. Wu, “Unet 3+: A full-scale connected
UNet for medical image segmentation,” 2020. [Online]. Available: